Document Type : Original Article
Authors
Department of Physics, College of Science, University of Baghdad, Baghdad, Iraq
Abstract
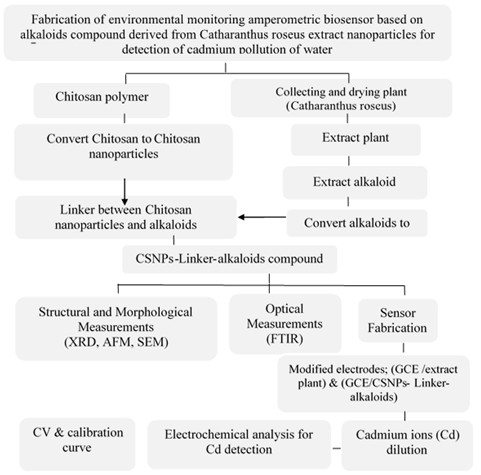
In this work, the Soxhlet extraction method extracted the Catharanthus roseus plant to obtain alkaloids, and then converted them into Chitosan nanoparticles (CSNPs)-linker-alkaloids compound. The CSNPs- linker-alkaloids compound was characterized by X-ray diffractometer (XRD), which shows the sample has an orthorhombic structure with crystallite size in nano-dimension; in Fourier Transform Infrared spectroscopy (FTIR), a new distinct band appears at 1708.93 cm-1 which is attributed to C=O esterification, which indicates the success of the reaction. Scanning electron microscope (SEM) images show that the final product has two shapes, spherical particles and tubes in nano-dimensions, and is close to each other compared to normal Chitosan. The cyclic voltammetric technique was used to study the electrochemical analysis of the newly modified electrode with extract plant and CSNPs-linker-alkaloids compound on the glassy carbon electrode. Citrate-Phosphate buffer, KCl electrolytes at pH 5, 20 cycles, buffer volume to 1000 μl and 0.5 Vs-1, the extract plant, CSNPs-linker-alkaloids storage for six months conditions are considered. The modified electrode was used to detect cadmium (Cd) ions in the range of (0.02 and 0.16) ppm. The best was sensitivity R² equal to 0.98 for oxidation and 0.975 for reduction. The modified electrodes; (GCE/extract plant( and )GCE/CSNPs-linker-alkaloids) act as suitable sensors for heavy metals detection in water.
Graphical Abstract
Keywords
Main Subjects
Introduction
Catharanthus roseus (C. roseus) is a member of the family Apocynaceae, one of the most widely explored plants because of its ornamental values and medicinal properties. The plant has been popularly exploited because of its reservoir for more than 200 alkaloids [1, 2]. A biosensor is a device that measures biological or chemical reactions by generating signals proportional to the concentration of an analyte in the response. Biosensors are employed in applications such as environmental monitoring, disease monitoring, detection of pollutants, disease-causing micro-organisms, drug discovery, and markers of disease in bodily fluids (urine, blood, sweat, and saliva) [3, 4]. Biosensors used for environmental have several advantages over conventional systems and methods, some of which are: their ability for portability, miniaturization and measurement of a pollutant with minimal samples. Most biosensors were developed to detect heavy metals, nitrogen compounds, pesticides, herbicides, dioxins, and phenolic compounds. As displayed in Figure 1 [5], some environmental biosensors exist. Sources of heavy metals in the environment are released from natural weathering processes of the earth’s crust, industrial discharge, soil erosion, sewage effluents, air pollution, mining, urban runoff, and control agents applied to plants, among other sources. The natural sources of soil leaching and metals in waters are the chemical weathering of minerals [6]. The highly toxic material of heavy metal in the environment is Cadmium (Cd). Cadmium gets into water and soil from waste discharge and leaches at dangerous waste sites. Cadmium can be released into the atmosphere in several ways: biological activities, such as volcanic activity weathering and erosion, river transport, and human activities, such as fossil fuel combustion, tobacco smoking, smelting, mining, incineration of municipal waste manufacture of phosphate fertilizers, recycling of cadmium-plated steel scrap as well as electric and electronic waste. Cadmium exerts toxic effects on the kidney, the skeletal, and respiratory systems and is classified as a human carcinogenic. Cadmium at high levels in foods or water can cause stomach irritation, high blood pressure, vomiting, nausea, convulsions, diarrhea, liver disease, headache, brain damage, weakness, fever, and kidney problems, which sometimes lead to death [7]. This work is aimed to fabricate amperometric biosensors based on eco-materials (plants) to detect traces of heavy metals such as Cadmium ions (Cd) which cause environmental contamination, such as water pollution, by measuring the cyclic voltammetry by two types of modified glassy carbon electrodes with; extract plant and CSNPs-linker-alkaloids compound.

Water pollution is the primary form of pollution as chemical pollutants enter rivers, lakes, and wetlands. Toxic chemical substances introduced into the environment may be transported by water, air, and living organisms, become a part of the natural biogeochemical cycle, and accumulate in the food chain. Water constitutes the “trouble spot” of all ecosystems, as many pollutants are waterborne. It also plays a vital role as a solvent for various substances and a medium in the cycle of air-soil-plants-animals [8].
Materials and Methods
Chemicals and reagents
The chemical materials that are used in this work are Catharanthus roseus (C46H56N4O10) (origin from Madagascar, planted in Iraq), Chitosan (C6H11NO4)n of purity 98% from Weifang Dongxing Shell Products Factory, (Korea), Sodium TriPolyPhosphate (STPP) (Na₅P₃O10) of purity 94% from Andhra Pradesh, (India), Maleic anhydride (C4H2O3) of purity 98% from Sinopharm Chemical Co., Ltd., (China), and Tetrahydrofuran (THF) (C₄H₈O) of purity 99% from Sigma-Aldrich. For electrolyte solution: Potassium chloride (KCl) of purity 99 % from Sigma-Aldrich, Sodium chloride (NaCl) of purity 99% from British Drug Houses (BDH), Ltd. Pool, (England), sodium carbonate (Na2Co3) of purity 99 % from Sigma-Aldrich. For Buffer: Citric acid (HOC(COOH)(CH2COOH)2) of purity 99.5%, sodium citrate (HOC(COONa)(CH2COONa)2.2H2O) of purity 99%, acetic acid (CH3CO2H) of purity 99%, sodium acetate (CH3COONa) of purity 99%, and dibasic sodium phosphate (dihydrate) (Na2HPO4·2H2O) of purity 99%. All these materials were from Sigma-Aldrich, and heavy metal: Cadmium (Cd) stock solution with concentration (1000 ppm) from Horiba, (France).
Instrumentation
An electrochemical system (IRASOl PGS-10) with potentiostats driven by electroanalytical measuring software was connected to a computer to perform the CV used in the experiment. The electrochemical response was determined by calculating the redox current peaks with dependence on parameters. All measurements were carried out in a three-electrode cell containing a glassy carbon electrode (GCE) as the working electrode (WE), Ag/AgCl potassium chloride as a reference electrode (RE), and platinum wire as a counter electrode (CE) [9].
Preparation of CSNPs-linker- alkaloids
Catharanthus roseus is cultivated in Iraq. The plant was collected and dried at room temperature (R.T.), and then it was ground by an electric grinder. The Soxhlet method was used to extract Catharanthus roseus. Soxhlet apparatus has an extractor volume of 85 mL, and a flask volume of 250 mL from Sigma-Aldrich. The extraction temperature was 50 °C, this temperature considers the best for extracting alkaloid compounds which do not destroy or change them [10]. Figure 2 indicates the schematic diagram of Catharanthus roseus alkaloids extracts.
Chitosan was used to synthesize Chitosan nanoparticles (CSNPs) in the presence of Sodium TriPolyPhosphate (STPP) compound as a correlation factor with the Chitosan to produce the Chitosan nanoparticles during the inotropic gelation method. The preparation process and the ratios were used according to the plan reported in [11]. Figure 3 depicts the schematic diagram of CSNPs-linker-alkaloids preparation.


A weight of 3 g of the prepared Chitosan nanoparticles (CSNPs) has been taken and solved into 10 ml of Tetrahydrofuran (THF), put into a cold-water bath, and subjected to ultrasonic for 5 min. using the probe sonicate. The required concentration of the alkaloid extract was prepared by taking 1 g of it, melting it in a volume of (THF), and using a sonicated probe for 5 min. to reach the alkaloid extract to nanoscale size.
The alkaloid extract was linked to the chitosan nanoparticles compound using a similar approach to what was mentioned with some modifications. Alkaloid extract was taken and dissolved well in Tetrahydrofuran (THF), and then subjected to ultrasonic for 5 min. using the probe sonicate device. It was added by distillation to the Chitosan nanoparticles solution with the presence of maleic anhydride as a connection between them under a magnetic stirrer for two hours at 45 °C to obtain the resulting component form (CSNPs-linker-alkaloids) as a nano-powder [11, 12].
Working electrode cleaning and modification
The GCE was polished with 0.05 μm alumina powder using emery papers, rinsed with deionized water, and then cleaned with acetone by ultrasonication for 5 min.
Perpetration of the heavy metal
The maximum concentration of the heavy metal element (Cd) was prepared from the stock solution. The necessary dilution was done with distilled water based on the following chemical formula [13].
![]()
Where, C1 is the concentration of a stock solution (ppm), V1 is the volume obtained from the stock solution (mL), C2 is the final concentration (ppm), and V2 is the final volume (mL).
Results and Discussion
The structure of the CSNPs-linker-alkaloids was studied using an X-ray diffractometer (ADX2700Angstrom model XRD-6000 USA, 40 VK, 30 mA, λ1.54 Å). The patterns of the sample are displayed in Figure 4. The characteristic peaks of CSNPs-linker-alkaloids compound is at a diffraction angle of 2θ=22.3° corresponding to (101) planes, which agrees with the results of [14]. The synthesis sample shows the orthorhombic structure, which blends with Okuyama et al. [15]. Previous researchers inferred the appearance of the broad peak at a diffraction angle of 22.3° [16]. As a result of the arrangement of the CSNPs-linker-alkaloids compound chains in the form of parallel planes to give the orthorhombic form [17].

Scherrer’s Equation [18, 19] was used to calculate the crystal size (C.S) of the synthesized CSNPs-linker-alkaloids compound:
C.S = (K' λ)/ (β' Cos θ) (2)
Where, β’ is the angular line width at half maximum intensity equal to 7ᵒ according to the XRD results, K' is a constant with a value of 0.9, and θ is the Bragg’s angle C.S of the CSNPs-linker-alkaloids is 1.2 nm.
Figure 5 exhibits the spectrum of CSNPs-linker-alkaloids. The spectrum shows a wide band at 3344.56 cm-1 which is attributed to chitosan O-H. Similarly, the unreactive O-H in Vintafolide is one of the added alkaloid compounds as it contains more than one O-H group, so the peak appeared somewhat wide as this compound contains more than five non-reactive OH compounds, but in addition to other groups reactive with the compound for that more than one peak appears in the spectrum. The spectrum appeared C-H (aromatic) bond at 3150 cm-1. The spectrum appears to peak at 2904.79 cm-1 and 2855.04 cm-1 corresponding to C-H symmetric and asymmetric stretching vibration stretch (C-H). In addition, this spectrum shows a new distinct band at 1708.93 cm-1 due to C=O esterification, which indicates the success of the reaction as the carboxy turns into esterification and its frequency shifted to higher than it is in Chitosan with the maleic anhydride. The peaks at 1627.92 cm-1 and 1527.26 cm-1 are attributed to the NH group, while the peak at 1459.04 cm-1 is attributed to CH (wagging for the CH = CH group). The peak at 1384.89 cm-1 is attributed to the P=O overlap with the C-O vibration stretch. The peaks at 1087.85 cm−1, 1018.41 cm-1, and 875.68 cm−1 are due to the presence of C–O stretching, P–O, or PO4, and C-H bending(wagging).
According to Figure 5, the synthesis process is successful and this is due to the width of the bundles and the groups of active alkaloids, which causes the bundles to interfere with each other, and a new distinct band appears at 1708.93 cm-1.
Scanning electron microscope (SEM) images give the film's crystallographic, composition, and topographic. A Jsm-7610F scanning electron microscope was used to observe the structure of CSNPs-linker-alkaloids compound as shown by SEM images in Figure 6. When examining the Chitosan nanoparticles (CSNPs) with the extracted plant, it was found that two shapes in nano dimensions were formed (spherical particles and tubes), as appeared in Figures 6a,b.

They are regular and have dimensions with a diameter range of 13-44 nm. These spherical particles and tubes are close to each other, which proves the success of synthesis of material (CSNPs-linker-alkaloids compound) when adding the binding material (the maleic). The success of linking alkaloids with Chitosan nanoparticles (CSNPs) by the maleic gives a new compound that can interact with alkaloids, and this is through electronic pairs of hydrogen bonds, as it works on the convergence of chitosan nanotubes. It is also noted from the figure that nanoparticles of the new compound became larger than the Chitosan nanoparticles (CSNPs).
The images represent that the synthesis CSNPs-linker-alkaloids compound has two shapes in nano dimensions; spherical particles and nanotubes due to nuclei and growth processes, respectively.
Study the optimal condition for extracted plant, CSNPs-linker-alkaloids compound
It is possible to summarize the best conditions and their effect on the oxidation and reduction peaks used in cadmium metal ions detection applications. The best conditions were chosen to prepare the pollutants of water to be measured by the CV technique.
Effect of electrolyte’s pH
The cyclic voltammetric study was performed in potassium chloride solution with different pH (1-9) of the (GCE/ extract plant), and (GCE/ CSNPs-linker-alkaloids compound). Figure 7 illustrates the effect of different electrolyte solution pH on the behavior of oxidation-reduction current peaks of an extract plant, and CSNPs-linker-alkaloids compound.
The modified electrode (GCE/ extract plant) and (GCE/ CSNPs-Linker-alkaloids compound) for acidic HCl and alkaline NaOH medium. The oxidation-reduction current peaks of the modified electrodes (GCE/ extract plant), and (GCE/ CSNPs-Linker-alkaloids compound) increased with the solution pH in high acidic values pH 2-5, and then decreased in pH 6-9. The optimum oxidation-reduction current peaks (best response) were detected for the acidity function 5 for both electrodes. This amount of acidity is the most suitable for charging the active groups, activating, and preparing them for the ions under study. Therefore, the response will be the highest possible and this acidity can be considered as a catalyst for the reaction.
In basic media (electrolyte), the response begins to decrease and then becomes zero in high basic media, because the ester bond will dissolve to give the carboxylic salt and the corresponding alcohol. The salts of carboxylic acids and amines in the alkaloids will be positively charged, because of the presence of a large number of negative ions, the reaction will be a salt formation reaction, which does not allow the positive ions to respond [20].
Effect of the buffer type
Buffers are aqueous systems that resist changes in pH when small amounts of acid or base are added. It is necessary to use the appropriate buffer solution to maintain the pH level. The working electrode tends to change the pH solution level at the surface layer. This has an impact on the reduction voltage and results in weak peaks. Thus, a non-linear relationship is expected between diffusion current and KCl concentration. Therefore, the solution should be properly regulated in the organic substance cycle voltammetry to reach a controlled quantitative and qualitative level [21].
Alkaloids compound in different buffer types; Citrate-phosphate, citrate, and acetate at a concentration of 16500 ppm of extract plant, and CSNPs-linker-alkaloids have a pH value of 5. According to Figure 8, the best buffer type is citrate-phosphate for both modified electrodes.


Effect of the buffer volume
Buffer capacity is a term used to describe the ability of a given buffer to resist changes in the pH value to the addition of acid or base. The effect of buffer volume was investigated in the range of 500-2500 μl.
Figure 9 illustrates the results of the cyclic voltammogram for different buffer volumes. The diffusion current increases upon increasing the buffer volume up to 1000 μl for each extract plant, and CSNPs-linker-alkaloids modified electrodes. When the volume increases, the current decreases or the sensitivity decreases. This is due to the possibility of changing the substance shape to be measured. CSNPs-linker-alkaloids give better than that obtained currently from extract plants.
Effect of the electrolyte type
An electrolyte solution is electrically conductive and involves ions. atoms, or molecules that have lost or retained electrons.
They are sometimes referred to as ionic solutions, though there are some situations where the electrolytes are not ions [22]. The electrolytes effect was studied using three types; potassium chloride (KCI), sodium chloride (NaCl), and sodium carbonate (Na2CO3) at pH 5, with the dependence of citrate-phosphate as a buffer. Figure 10 represents the effect of electrolyte type on the oxidation-reduction current of the extract plant and CSNPs-linker-alkaloids. Experiments have proven that potassium chloride is the best among the selected electrolytes; this could be because both its negative and positive ions have the same mobility speed because of their very close conductivity, unlike the rest of ions in other salts [23]. Potassium chloride (KCI) is better than sodium chloride (NaCl), and sodium carbonate (Na2CO3). Therefore, the KCl electrolyte gives the highest response and the highest electrochemical stability [24].
Stability of extract plant and CSNPs-linker-alkaloids
Long-term stability is an important parameter in the real application of biosensors. Thus, the stability of the extract plant, CSNPs-linker- alkaloids biosensor was investigated by examining its CV peak current of optimum electrode conditions using 20 cycles. The used extract plant, CSNPs-Linker-alkaloids were stored for 6 months. The study was carried out at room temperature, KCl electrolytes of pH 5, citrate-phosphate buffer, and a scan rate of 0.5 Vs-1.

The cyclic voltammogram in Figure 11 proves that the extract plant and CSNPs-linker-alkaloids compound have chemical stability.
Effect of the scan of an extract plant, CSNPs-linker- alkaloids taken under optimum conductions
The previous optimum conditions were used to study the effect of scan rate in the range of 0.02-0.5 Vs-1 on the CV curve. According to Figure 12, the oxidation-reduction current increased linearly with increasing the scan rates, where the diffusion process at slow voltage scan rates grows much more on the electrode surface than at fast scan rates. Therefore, at fast scan rates, the flux of ions to the electrode surface is much larger than it is at slow scan rates. As the current is proportional to the flux, the current intensity becomes lower at slow scan rates and higher at high scan rates. This means that the electrode transfer reaction (the redox system) was diffusion-controlled. Thus, the modified electrode acts as an electro-catalyst with the presence of extract plant and CSNPs-linker-alkaloids compound.
Detection of heavy metals (Cd) by cyclic voltammetry
One of the most severe environmental issues is heavy metal contaminants. Industrial waste can damage the nervous system because it contains cadmium (Cd) ions. Figure 13 demonstrates the cyclic voltammograms of the extract plant and CSNPs- Linker-alkaloids modified glassy carbon electrodes used to study the detection of cadmium (Cd) ion at a concentration of 0.1 ppm (the maximum allowed concentration of heavy metals in rivers and water from pollution and is recommended for the system of maintaining rivers from pollution and according to Iraqi specifications No. (25 for 1967) [25].

Figures 14 and 15 appeared that at a concentration of 0.02 ppm, there is no current (the sensor does not respond), while at the range of 0.04-0.16 ppm, the oxidation and reduction current peaks enhance with increasing concentration.
The calibration graphs in Figure 16 display the relation between the oxidation-reducing current peaks resulting from CV and the concentration of 0.02-0.16 ppm for Cd metal.


Conclusion
Catharanthus roseus plant was successfully extracted and converted into Chitosan nanoparticles. The XRD results prove that the synthesized samples have an orthorhombic structure with crystal size in nano-dimensions. The FT-IR spectrum for CSNPs-Linker-alkaloids appear a new distinct band at 1708.93 cm-1, which is attributed to C=O esterification, which is evidence of the success of the synthesis process. The SEM images prove that the CSNPs- Linker- alkaloids formed in two shapes; spherical particles and tubes in nano-dimensions. Citrate-Phosphate buffer, KCl electrolytes at pH 5, 20 cycles, buffer volume to 1000 μl and 0.5 Vs-1, the extract plant, CSNPs- Linker- alkaloids storage for six months are found to be convenient for detecting Cadmium metal. The fabricated sensors are efficient for detecting water pollution with traces of heavy metals such as Cadmium metal. Therefore, the electrochemical sensor can be applied to quantitatively determine heavy metal ions in natural water samples with satisfactory results.
Acknowledgments
The authors thank Dr. Ameer Faisal, College of Science, University of Baghdad. The staff of the Environment and Water Directorate, Ministry of Science and Technology, and Prof. Bushra M. J. Alwash, Biology Department, College of Science for Women, University of Baghdad, for their help and support throughout the work.
Funding
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Authors' contributions
All authors contributed to data analysis, drafting, and revising of the paper and agreed to be responsible for all the aspects of this work.
Conflict of Interest
We have no conflicts of interest to disclose.
Orcid
Wasan R. Saleh
https://orcid.org/0000-0003-1321-5740
HOW TO CITE THIS ARTICLE
Marwah A. Al-Azzawi, Wasan R. Saleh. Fabrication of Environmental Monitoring Amperometric Biosensor Based on Alkaloids Compound Derived from Catharanthus Roseus Extract Nanoparticles for Detection of Cadmium Pollution of Water. Chem. Methodol., 2023, 7(5) 358-371